 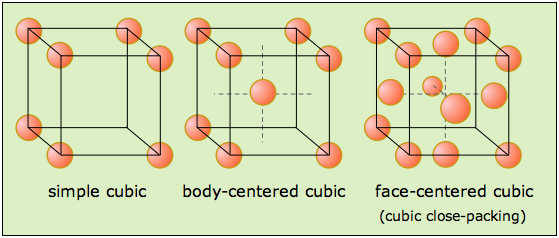
In the Mannheim process, sodium chloride is used for the production of sodium sulfate and hydrochloric acid. Sodium carbonate, in turn, is used to produce glass, sodium bicarbonate, and dyes, as well as a myriad of other chemicals. Sodium chloride is used in the Solvay process to produce sodium carbonate and calcium chloride. Sodium hydroxide is extensively used in many different industries enabling production of paper, soap, and aluminium etc. Some applications of chlorine include PVC thermoplastics production, disinfectants, and solvents. Other technologies are under development due to the high energy consumption of the electrolysis, whereby small improvements in the efficiency can have large economic paybacks. Each of those uses a different method to separate the chlorine from the sodium hydroxide. This electrolysis is conducted in either a mercury cell, a diaphragm cell, or a membrane cell. 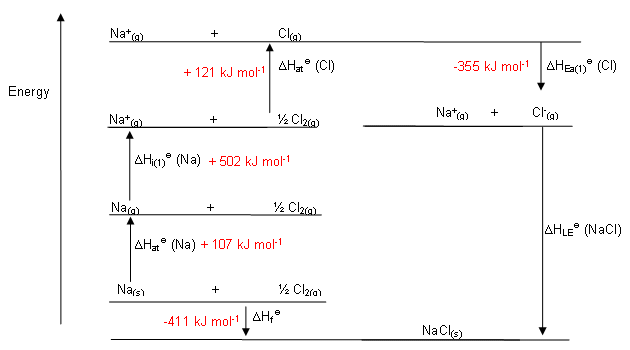
It does not store any personal data.It is the starting point for the chloralkali process, the industrial process to produce chlorine and sodium hydroxide, according to the chemical equation 2 NaCl + 2 H 2 O → e l e c t r o l y s i s Cl 2 + H 2 + 2 NaOH The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. 
The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Calculate the lattice enegry of CaCl2 from the given data.How do you find the lattice energy of CaCl2? We can substitute these values in the equation to find out the lattice energy. Standard Enthalpy of Formation of CaCl2 (s) = −795 kJ. 
How do you solve lattice energy problems? In 1918 Max Born and Alfred Landé proposed that the lattice energy could be derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. The Born–Landé equation is a means of calculating the lattice energy of a crystalline ionic compound. What is the lattice energy equation derived from? When calcium ion (Ca2+) combines with oxide ion (O2-), calcium oxide (CaO) crystal forms, and 3414 kJ of energy is released. Lattice Energy Examples When sodium ion (Na+) combines with chloride ion (Cl–), sodium chloride (NaCl) crystal forms, and 787.3 kJ of energy is released. The bond between ions of opposite charge is strongest when the ions are small. The lattice energy of NaCl, for example, is 787.3 kJ/mol, which is only slightly less than the energy given off when natural gas burns. ∆H4 = Electron affinity of Cl(s) = – 349 kJ mol-1.∆H3 = dissociation energy of Cl2(s) = 244 kJ mol-1.∆H2 = ionisation energy of Na(s) = 495 kJ mol-1.∆H1 = heat of sublimation of Na(s) = 108.7 kJ mol-1.∆Hf = heat of formation of sodium chloride = 411.3 kJ mol-1.How do you find the lattice energy of NaCl? Lattice energies cannot be measured directly but are obtained from a thermochemical cycle called the Born–Haber cycle, in which Hess’s law is used to calculate the lattice energy from the measured enthalpy of formation of the ionic compound, along with other thermochemical data. How do you find the lattice energy of a crystal? Instead, the lattice energy is calculated by subtracting the other four energies in the Born–Haber cycle from the net enthalpy of formation. The net enthalpy of formation and the first four of the five energies can be determined experimentally, but the lattice energy cannot be measured directly. How do you find the lattice energy of a Born-Haber cycle? This Lattice Energy Formula is as follows: U=−rack′Q_1Q_2r_0 U is always a positive number, and it represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. We can compute the lattice energy of nearly any ionic solid by using a modified form of Coulomb’s law. 24 Which has higher lattice energy NaCl or MgO? What is lattice energy calculate lattice energy? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
